 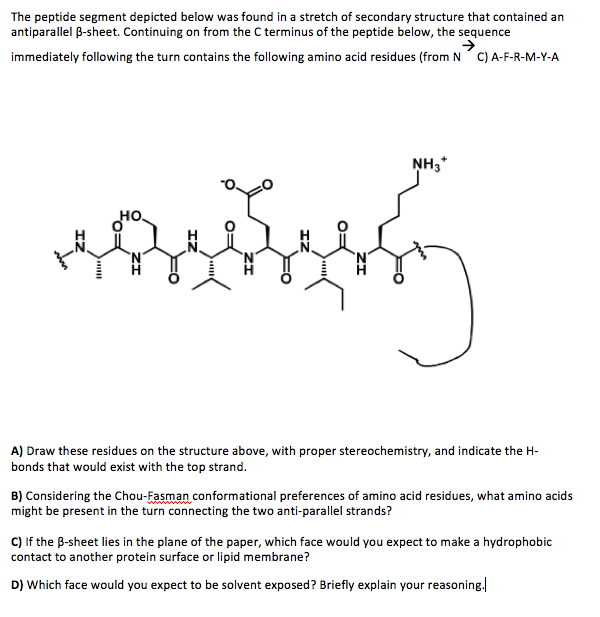
The C-terminal (96–140 aa) region of α-Synuclein is rich in acidic residues, which confer a net negative charge and a random coil structure. The exact binding mechanism is not fully understood, although a recent study suggested that oligomers can create membrane pores 11 or stabilise pre-existing membrane defects 12. 
The binding of misfolded states of α-Synuclein to lipid membranes is believed to be crucial for the development of PD and DLB. Although it is defined as an intrinsically disordered protein, previous structural investigations revealed that α-Synuclein folds into a helical structure, thereby following an apolipoprotein-type A2 helix mechanism, when bound to SDS micelles 9 and phospholipid vesicles 10. The α-Synuclein protein consists of 140 amino acids, is generally natively unfolded, and has lipid binding properties due to four amphipathic and conserved 11-repeats (KTKEGV) of the net positively charged N-terminal region (1–60 aa). The exact physiological role of α-Synuclein is not well understood, although reports suggest that it is involved in neurotransmitter regulation 7, 8. However, in vitro and in vivo results suggest that intermediate pre-fibrillar soluble forms, such as oligomers and protofibrils of α-Synuclein, possess more cell degenerating properties than insoluble Lewy body inclusions 2, 3, 4, 5, 6. The major constituent of Lewy bodies is the misfolded and fibrillated α-Synuclein protein 1, which has an important part in the pathogenesis of PD and DLB. These disorders are associated with neuropathological hallmarks such as loss of neurons in certain parts of the brain accompanied by intracellular deposition of Lewy bodies and Lewy neurites in surviving neurons. Neurodegenerative disorders such as Parkinson’s disease (PD) and dementia with Lewy bodies (DLB) are becoming more common probably as life expectancies are increasing. Due to its expected toxicity, this β-sheet motif may be a promising molecular target for the development of therapeutic strategies for PD and DLB. The results also suggest that early aggregated forms of the capped NAC 71–82 peptide generated structures were stabilised by an anti-parallel and twisted β-sheet motif. These results shed light on the aggregation propensity of the NAC 71–82 amino acid stretch of the full-length protein but also the roles of the N- and C-terminal domains of α-Synuclein in balancing this aggregation propensity. Here, we provide evidence that fibrils prepared from an acetylated and methyl amidated peptide of the NAC 71–82 amino acid stretch of α-Synuclein are amyloid and contain, in addition to the cross-β structure detected in the full-length protein fibrils, a cross-β structure previously observed in prion proteins. Although reports have demonstrated the importance of this fragment for the aggregation properties of the full-length protein, its exact role in pre-fibrillar oligomerisation, fibrillar growth and morphology has not yet been fully elucidated. The 71–82 fragment of the non-amyloid-β component (NAC) region of the Parkinson’s disease (PD) and dementia with Lewy bodies (DLB) related protein α-Synuclein, has been reported to be important during protein misfolding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
